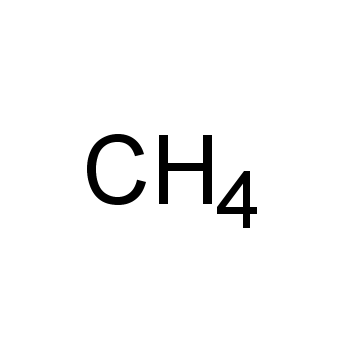
Is CH4 Polar? Uncovering the Mysteries of Methylene Gas
Is CH4 Polar? Uncovering the Mysteries of Methylene Gas
The world of chemistry is filled with fascinating molecules, each with its unique properties and characteristics. One such molecule that has garnered significant attention in recent years is methane (CH4), also known as methylene gas. As a simple hydrocarbon, CH4 is the primary component of natural gas, playing a crucial role in the global energy landscape. But is CH4 polar? To answer this question, we'll delve into the intricacies of molecular polarity, exploring the factors that contribute to a molecule's overall polarity. From the electron distribution to the molecular shape, our journey will uncover the secrets behind CH4's polarity.
Polarity in molecules is a result of the uneven distribution of electrons, which leads to a temporary dipole moment. This dipole moment arises from the difference in electronegativity between the atoms within a molecule. Electronegativity is a measure of an atom's ability to attract and hold onto electrons. The higher the electronegativity of an atom, the greater its ability to attract electrons.
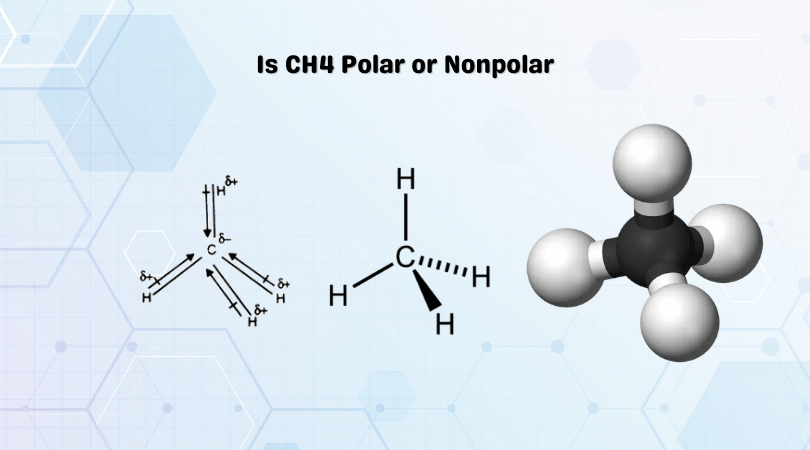
The molecular structure of methane consists of one carbon atom bonded to four hydrogen atoms in a tetrahedral arrangement. This structure seems to indicate that methane should exhibit some degree of polarity, given the difference in electronegativity between carbon and hydrogen. However, upon closer inspection, we find that the electronegativity values for carbon and hydrogen are relatively close, with carbon having an electronegativity of 2.55 and hydrogen having an electronegativity of 2.20 on the Pauling scale.
Despite the relatively small difference in electronegativity, CH4 exhibits some degree of polarity due to the molecular shape and the localized charge distribution along the C-H bonds. The tetrahedral geometry of methane leads to a symmetrical distribution of electron density around the carbon atom, resulting in a slightly negative charge on the carbon atom and a slightly positive charge on the hydrogen atoms.
However, this polarity is not enough to make CH4 a highly polar molecule. In fact, methane is considered a non-polar molecule due to the very low dipole moment arising from its bond dipoles. Each C-H bond in CH4 has a dipole moment of about 0.405 debye, which is relatively small compared to other molecules. The dipole moments of the four C-H bonds in CH4 are largely canceled out due to the tetrahedral geometry, resulting in a negligible overall dipole moment.
As a result, methane exhibits properties typical of non-polar molecules, such as low solubility in water and high boiling point.
Understanding Molecular Polarity
Molecular polarity can be understood through the following factors:
* **Electronegativity difference**: A larger difference in electronegativity between atoms results in a greater polarizing effect.
* **Bond length**: Shorter bond lengths, such as those found in polar covalent bonds, often lead to greater polarity.
* **Molecular shape**: Molecules with symmetrical shapes tend to exhibit reduced polarity, while asymmetrical shapes can result in increased polarity.
* **Atomic size**: Larger atoms tend to exhibit lower electronegativity and reduced polarity.
The Implications of CH4's Polarity
CH4's non-polar nature has significant implications for various fields, particularly in the domains of chemistry and physics. For instance:
* **Solubility**: Non-polar molecules like CH4 exhibit limited solubility in polar solvents, such as water.
* **Boiling point**: The relatively high boiling point of CH4 arises from its non-polar nature, which results in weaker intermolecular forces.
* **Chemical reactions**: CH4's non-polarity influences its reactivity in chemical reactions, often requiring specific catalysts or conditions to facilitate bond formation or breaking.
In conclusion, while CH4 exhibits a degree of polarity due to the localized charge distribution along the C-H bonds and the tetrahedral geometry, its negligible overall dipole moment and symmetrical electron distribution classify it as a non-polar molecule. The factors contributing to CH4's polarity offer insights into the principles of molecular polarity and its significance in various fields. By exploring these aspects, we can gain a deeper understanding of the intricacies involved in determining the polarity of molecules and its practical applications.



Related Post
Is CH4 Polar? Understanding Methane's Polarity

Is CH4 Polar or Nonpolar? The Shocking Truth About Methane Molecules

Is CH4 Polar or Nonpolar: A Simple Explanation

Amy Adams' Husband Unveils the Hidden Side of a Hollywood Icon's Marriage

