Is CH4 Polar? Understanding Methane's Polarity
Is CH4 Polar? Understanding Methane's Polarity
Methane, a potent greenhouse gas, is a vital component of the Earth's atmosphere, comprising about 1.7 parts per million (ppm). The Scientific American reports that methane's concentration has tripled since pre-industrial times, primarily due to human activities such as agriculture, natural gas systems, and land use.
Understanding the molecular structure of methane is crucial in assessing its implications for climate change. Methane (CH4) is a simple organic molecule composed of one carbon atom bonded to four hydrogen atoms in a tetrahedral arrangement. Its molecular structure is symmetrical, which often leaves scientists wondering: is methane polar?
Research indicates that polar molecules are those with a dipole moment, an electric dipole in which the molecule's center of positive charge does not coincide with its center of negative charge. A molecule's polarity can be affected by the difference in electronegativity between the atoms in the molecule. Electronegativity, or the ability of an atom to attract electrons in a covalent bond, varies across the periodic table. In the case of methane, the difference in electronegativity between carbon and hydrogen is not sufficient to generate a dipole moment, suggesting that methane has a much lower polarity.
The polarity of a molecule has substantial implications for its intermolecular interactions and, by extension, its thermodynamic properties. For instance, polar molecules are more prone to dispersion forces, also known as van der Waals forces, which cause them to stick together. In contrast, nonpolar molecules, like methane, rely on London dispersion forces, which are more non-directional and weaker than dispersion forces. These differences affect the boiling points and melting points of molecules and the rates of chemical reactions, among other characteristics.
Is Methane Polar: A Brief Look into the Foreground Chemistry
When it comes to the polarity of methane, we have to consider its chemical properties, particularly its reactivity and solubility. Polarity has a significant impact on the solubility of a substance. Polar molecules are more soluble in polar solvents due to greater intermolecular forces. However, methane's nonpolar nature limits its solubility in water, making it a poor solvent for this molecule.
Typically, nonpolar solvents are selected for separating components of a mixture because of their miscibility with nonpolar organic compounds. Furthermore, the polarity of a molecule affects its volatility. Volatile compounds can interact more easily with air because of their nonpolar characteristics. Methane, as a nonpolar molecule, is notably volatile and can interact easily with the atmosphere.
Understanding the Molecular Structure of Methane in a Nutshell
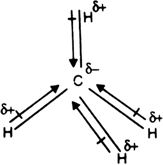
In order to grasp the issue of methane's polarity better, it's helpful to familiarize yourself with its structure by drawing a sample molecular layer. Methane's molecular structure consists of a central carbon atom bonded to four hydrogen atoms. The representation can also indicate the direction of bond dipoles. Viewing this image clears up all of the facts regarding its polar properties.
The unique tetrahedral arrangement of methane molecules can result in an overall dipole of zero. This enables carbon-hydrogen compounds to display unique properties. Each methane molecule has a C-H covalent bond, making hydrogen's electronegativity not sufficient to give the methan molecule an overall dipole.



Related Post

Unpacking Patrick Muldoon's Romantic History: A Comprehensive Guide to the Actor's Most Scandalous Moments

The Epic Puzzle: Unraveling the Mystery of Denver Post Mini Crossword

Hong Kong Stock Exchange: A Hub for Asia's Economic Growth

Extreme Weather Patterns: How Chris Bailey Kentucky Weather Blog Offers a Glimpse into the Complex World of Kentucky's Climate

