Is CH4 Polar or Nonpolar: A Simple Explanation
Is CH4 Polar or Nonpolar: A Simple Explanation
Imagine a molecule made of one carbon atom bonded to four hydrogen atoms, looking like a neat little tetrahedron. This molecule is methane (CH4), a simple yet vital compound found in natural gas. But is methane a polar molecule, or is it nonpolar? To understand the nature of methane's polarity, we need to delve into the world of chemistry and explore the properties of its molecular structure.
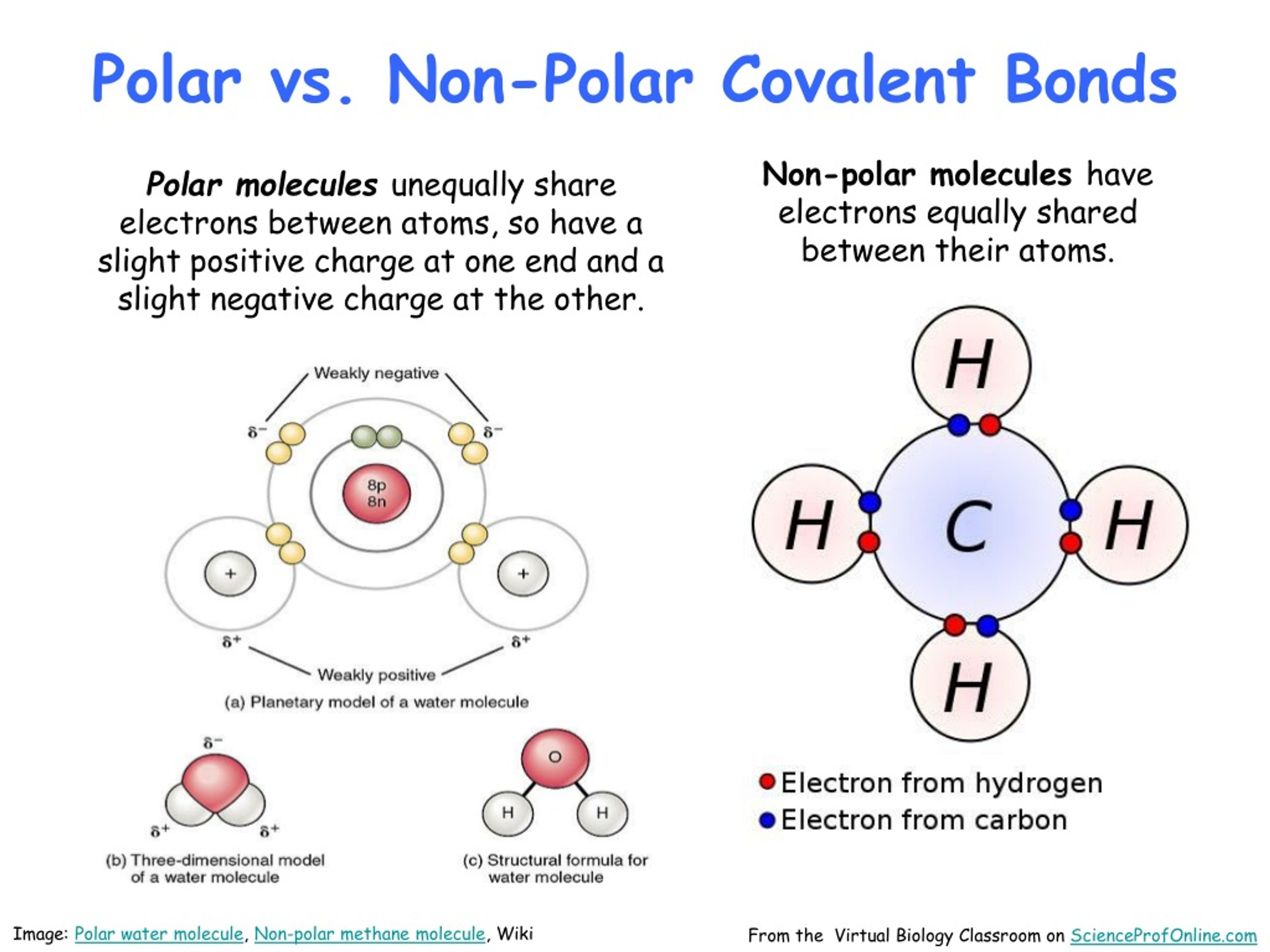
Methane's molecular structure consists of a central carbon atom bonded to four hydrogen atoms, all single bonds. The carbon atom has a slightly positive charge, while the hydrogen atoms have a neutral charge. However, the carbon atom is also bonded to the four hydrogen atoms, which creates a slight difference in electronegativity between the two. The difference in electronegativity between the carbon and hydrogen atoms is 0.35, which may be small but significant enough to determine the polarity of the molecule.
CH4 (methane) is a nonpolar molecule. It is a symmetrical molecule with a tetrahedral shape, which means that the electrons are equally distributed among the bonds. This symmetry ensures that there is no net dipole moment; the partial positive and negative charges cancel each other out.
Understanding Polarity in Molecules
What is Polarity in Molecules?
Polarity in molecules arises when there is a difference in electronegativity between the atoms in a bond. This difference in electronegativity leads to a separation of charges in the molecule.
* In a polar bond, there is a more negative and a more positive end, creating an electric dipole moment.
* In a nonpolar bond, the electrons are equally distributed in a symmetrical molecule.
* A nonpolar molecule does not have a net dipole moment; the partial positive and negative charges are equivalent within the molecule.
Characteristics of Polar and Nonpolar Molecules
Differences Between Polar and Nonpolar Molecules
| Characteristics | Polar Molecules | Nonpolar Molecules |
| --- | --- | --- |
| Polar Covalent Bonds | Bonds with a difference in electronegativity (more than 0.5) | Bonds with equal or little difference in electronegativity (less than 0.2) |
| Molecular Shape | Asymmetrical | Symmetrical |
| Dipole Moment | Has a net dipole moment | Does not have a net dipole moment |
Why CH4 is Nonpolar
Reasons Why CH4 Remains Nonpolar
* **Symmetry**: Let's consider the carbon atom in the center who has four identical bonds to the hydrogen atoms around it. Due to the symmetrical arrangement, every bond angle is the same; the charges on each bond remain neutral.
* **Electronegativity**: As mentioned earlier, the difference in electronegativity between carbon and hydrogen is just 0.35, meaning there is minimal polar character in each bond.



Related Post

'Jones Zone' Jon Jones New Home: A Look Inside His Albuquerque Mansion

Unveiling the Mystique of Christopher Pettiet: A Life of Hollywood Whispers and Forgotten Dreams

Il mistero dei volpi italiane: scoprire il fascino di un simbolo nazionale

Uncovering the Truth Behind Arrest Mugshots in Brevard County, FL: A Comprehensive Look

