Unveiling the Atomic Secrets of Magnesium: How Many Valence Electrons Does it Possess?
Unveiling the Atomic Secrets of Magnesium: How Many Valence Electrons Does it Possess?
Magnesium, a silvery-white alkaline earth metal, is one of the most abundant elements in the Earth's crust and is a key component in many biological systems. With its unique chemical properties and physical characteristics, magnesium has numerous applications in various industries, including construction, transportation, and electronics. However, behind this metal's diverse uses lies a complex atomic structure, which is fascinating for those interested in chemistry and physics. In this article, we will delve into the world of atomic electrons to explore how many valence electrons magnesium possesses and the implications this has for its behavior as a chemical element.
The Concept of Valence Electrons
To understand the number of valence electrons in magnesium, it is essential to grasp the concept of valence electrons themselves. Valence electrons are the electrons in an atom's outermost energy level, which participate in chemical bonding with other atoms. The number of valence electrons an atom has determines its chemical reactivity and the types of bonds it can form. Valence electrons are crucial in understanding various chemical phenomena, such as the periodic table of elements, acid-base chemistry, and the principles of chemical bonding.
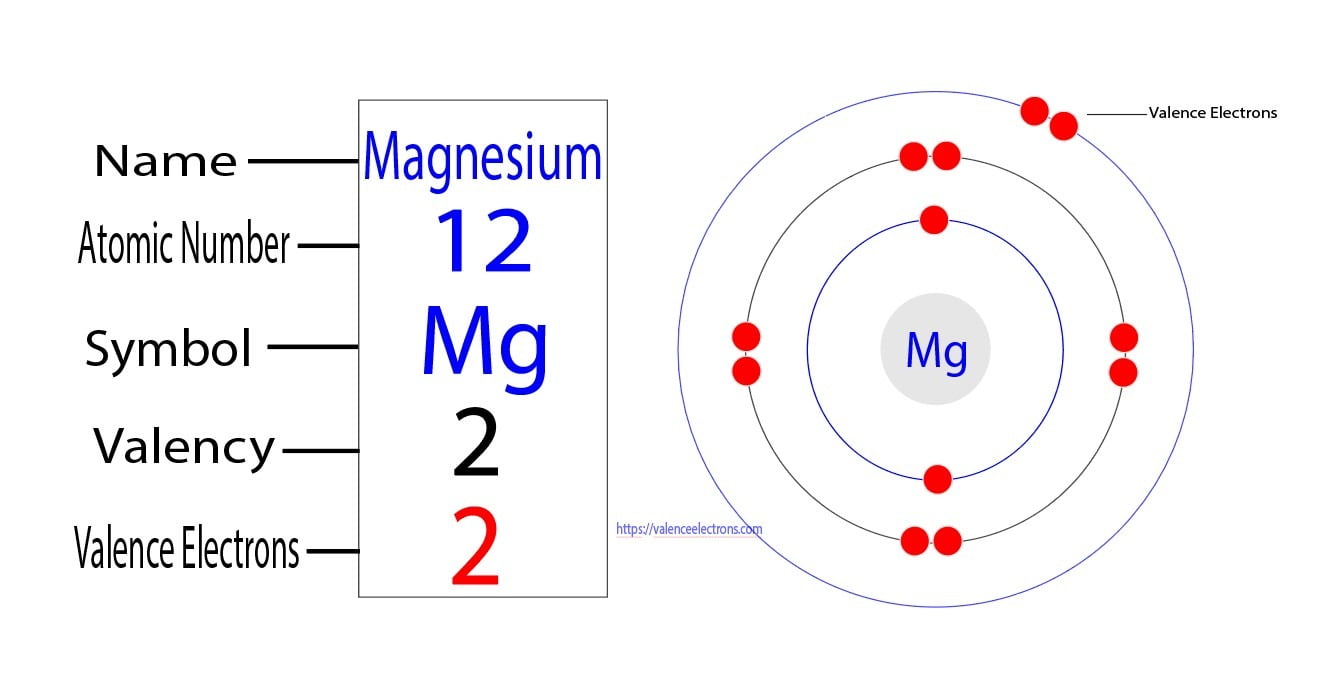
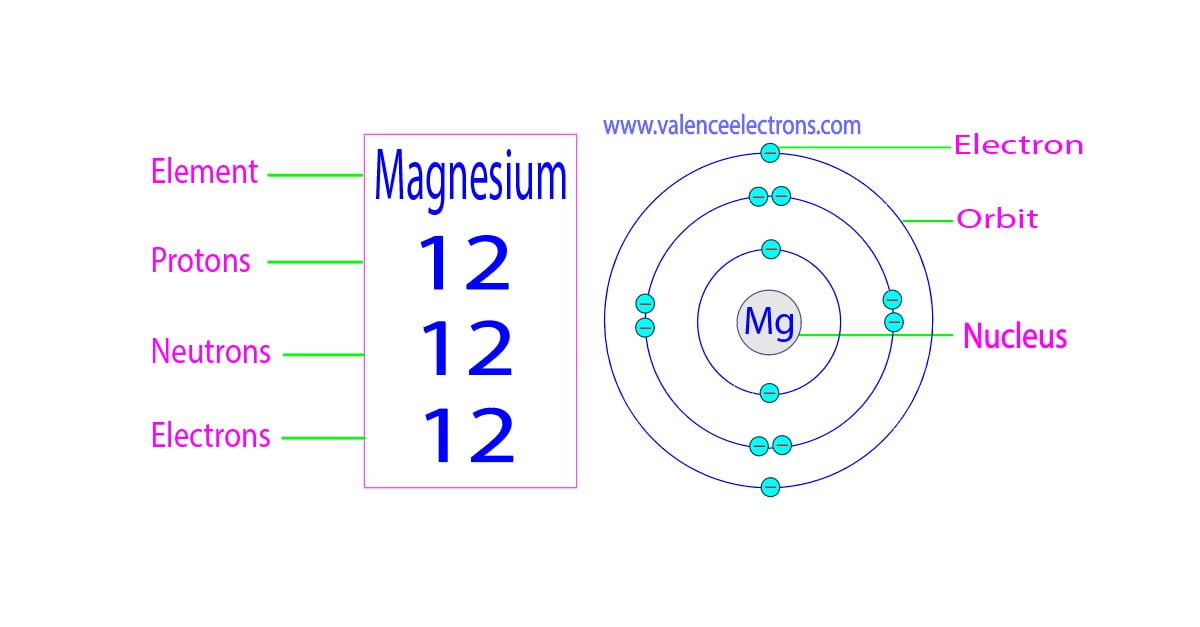
Magnesium, like other elements, has a specific atomic structure that consists of protons, neutrons, and electrons. The atomic number of magnesium is 12, indicating that a neutral magnesium atom contains 12 protons and 12 electrons. The electrons in an atom are arranged in various energy levels or shells, with the first two shells being filled with electrons before the next energy level begins. The electrons in the outermost shell are considered valence electrons, which are responsible for magnesium's chemical properties.
The Atomic Structure of Magnesium
To determine the number of valence electrons in magnesium, we need to examine its atomic structure in more detail. Magnesium has two electrons in its innermost energy level (1s^2) and one electron in its second energy level (2s^2). However, when considering valence electrons, we often focus on the electrons in the outermost energy level, which affect an atom's chemical reactivity. In the case of magnesium, the outermost energy level consists of two electrons in the 3s orbital, which are its valence electrons.
The electron configuration of magnesium can be written as 1s^2 2s^2 2p^6 3s^2. The superscripts denote the number of electrons in each orbital. In this arrangement, the 3s orbital contains two electrons, which are termed valence electrons. Therefore, magnesium has two valence electrons.
Implications of Valencia Electrons in Magnesium
Understanding the number of valence electrons in magnesium is crucial for predicting its chemical behavior and applications. Magnesium's two valence electrons make it highly reactive, as it tends to lose these electrons to form ions with a +2 charge. This is observed in magnesium's reaction with oxygen, where it also forms magnesia (MgO). The reactivity of magnesium is also the basis for its use in various chemical reactions, including the production of magnesium chloride (MgCl2) and magnesium hydroxide (Mg(OH)2).
The two valence electrons in magnesium also affect its physical and chemical properties. Magnesium has a relatively low melting point of about 649°C due to its tendency to lose electrons and form ionic bonds. Furthermore, the reactivity of magnesium has led to its use in various applications, including in fireworks, flares, and other pyrotechnic devices.
Conclusion and Real-World Applications
In conclusion, magnesium's two valence electrons play a significant role in its chemical behavior and numerous applications. Understanding the atomic structure and electron configuration of magnesium enables chemists and physicists to predict its reactivity and interactions with other elements. The significance of valence electrons in magnesium serves as an example of how atomic-level properties affect the behavior of elements and their uses in various industries.
By examining the atomic secrets of magnesium, we can appreciate the fascinating world of chemical bonding and the properties of elements, which underpin various technologies and scientific discoveries.

Related Post

Is Paula Patton Married? Unpacking the Actress's Personal Life and Relationships

Uncovering the Truth Behind the Bronx's Notorious Reputation: Is the Northernmost Borough of NYC a Danger Zone?
:max_bytes(150000):strip_icc():focal(599x0:601x2)/peter-mcmahon-dana-perino-instagram7-11424-c4fdddd0376f4ccdaf9117f11923e1be.jpg)
Dana Perino Wedding Pictures: A Beautiful Union of Love and Partnership

The Astonishing Transformation Cat Woman Plastic Surgery And Jocelyn Wildenstein's Journey 'Wildenstein Says She Never Had'