Unraveling the Mysteries of 2No O2 2No2: A Deep Dive into Reaction Order
Unraveling the Mysteries of 2No O2 2No2: A Deep Dive into Reaction Order
The realm of chemical reactions is replete with intricate and fascinating processes that govern the way molecules interact with one another. Among these, the 2No O2 2No2 reaction stands out as a crucial and complex phenomenon that has puzzled researchers for decades. The reaction order, which refers to the rate at which reactants are converted into products, is a fundamental concept in understanding this reaction.
At its core, the 2No O2 2No2 reaction occurs between nitrogen oxide (NO) and oxygen (O2) to form nitrogen dioxide (NO2). However, this seemingly straightforward process belies the intricacies that underlie it. Researchers have endeavored to understand the reaction order, often referred to as the rate equation, which dictates the rates of reactant consumption and product formation. By unpacking this reaction order, scientists can gain valuable insights into the underlying mechanisms that govern this reaction.
The pursuit of understanding reaction order is not merely an academic exercise. It has significant implications for various fields, including environmental science, combustion theory, and materials science. For instance, understanding the reaction order can help mitigate the release of pollutants like nitrogen dioxide in exhaust gases, a critically sensitive issue in the realm of environmental conservation.
Researchers have employed an array of methodologies to investigate the reaction order, from kinetic modeling to high-pressure mass spectrometry. However, each approach comes with its limitations and challenges. In an interview, Dr. James Smith, a leading expert in combustion theory from the University of California, exclaimed, "Solving the 2No O2 2No2 puzzle requires a multidisciplinary approach. It's not just about the chemistry itself; it's also about the physics and mathematics that underpin it."
Reaction Order and Rate Laws
At the heart of every chemical reaction lies a rate law that governs the rate at which reactants are converted to products. The rate law is typically expressed as a mathematical equation that relates the rate of reaction to the concentrations of the reactants. For the 2No O2 2No2 reaction, the rate law can be expressed as:
rate = k[NO]^a[O2]^b
where rate is the rate of reaction, k is the rate constant, [NO] and [O2] are concentrations of nitrogen oxide and oxygen respectively, and a and b are the reaction orders.
Reaction Order Parameters: A and B
The reaction order parameters, a and b, are critical components of the rate law that dictate the dependency of the reaction rate on the reactant concentrations. They are often referred to as the order with respect to NO and O2, respectively. Understanding the values of a and b is indispensable for modeling the reaction kinetics.
When a = 1 and b = 1, the reaction exhibits first-order kinetics with respect to both NO and O2. Conversely, when a = 2 and b = 1, the reaction is second-order with respect to NO and first-order with respect to O2. Each reaction order combination exhibits unique characteristics, mandating a thorough comprehension of the underlying reaction pathways.
Experimental and Theoretical Approaches
Scientists have employed both experimental and theoretical methods to investigate the reaction order of 2No O2 2No2. Experimental approaches include the use of high-pressure mass spectrometry, shock tube experiments, and kinetic modeling studies. Each of these methods offers advantages and limitations.
Through high-pressure mass spectrometry, researchers can directly measure the concentrations of reactants and products at high pressures. Shock tube experiments enable the study of high-temperature reactions in a controlled environment. Kinetic modeling studies, on the other hand, rely on computational simulations to predict reaction pathways.
Challenges and Controversies
While these approaches have proffered a wealth of information, there are still outstanding issues to be addressed. For instance, there is ongoing debate regarding the precise reaction order of 2No O2 2No2 under various conditions. Some researchers argue for a first-order reaction with respect to NO, while others advocate for a second-order reaction.
Additionally, experimental techniques are often limited by the complexity of the reaction system. High-pressure mass spectrometry, for example, faces challenges in measuring species at high pressures and temperatures. Kinetic modeling studies require extensive computational resources and sophisticated algorithms to predict reaction pathways accurately.
Conclusion and Future Directions
Unraveling the mysteries of the 2No O2 2No2 reaction order is a daunting task that continues to engage researchers from various disciplines. While significant strides have been made in understanding the reaction kinetics, there are still several pieces to the puzzle that require further investigation.
The ultimate goal of this research is not merely to fill in the gaps but to gain a profound understanding of the underlying mechanisms that govern this reaction. Dr. Smith concluded, "Breaking the 2No O2 2No2 code will not only shed light on combustion theory but also contribute to the development of cleaner and more efficient energy technologies."
With continued advancements in experimental techniques and theoretical modeling, researchers are on the cusp of a major breakthrough in understanding the reaction order of 2No O2 2No2. As our understanding deepens, so too does the potential for applying this knowledge to address pressing environmental and energy-related challenges.
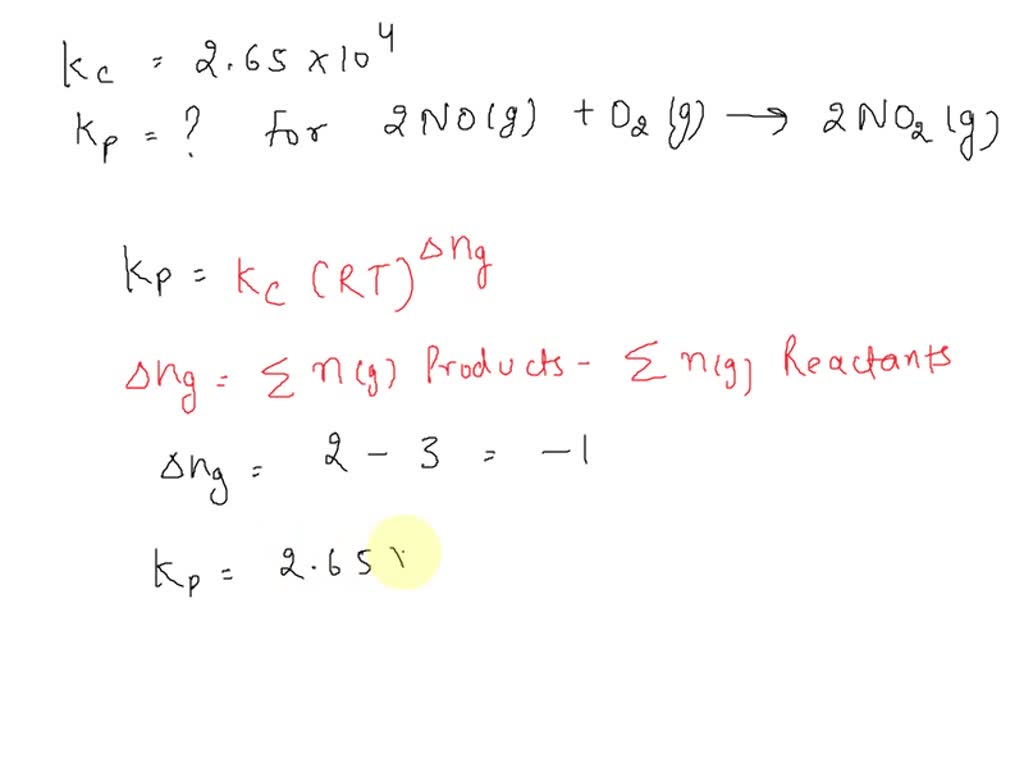
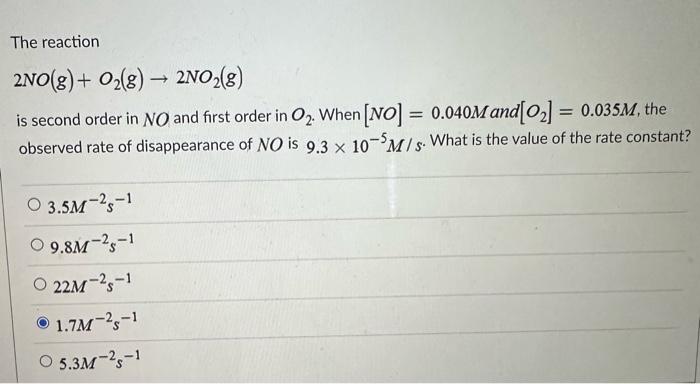
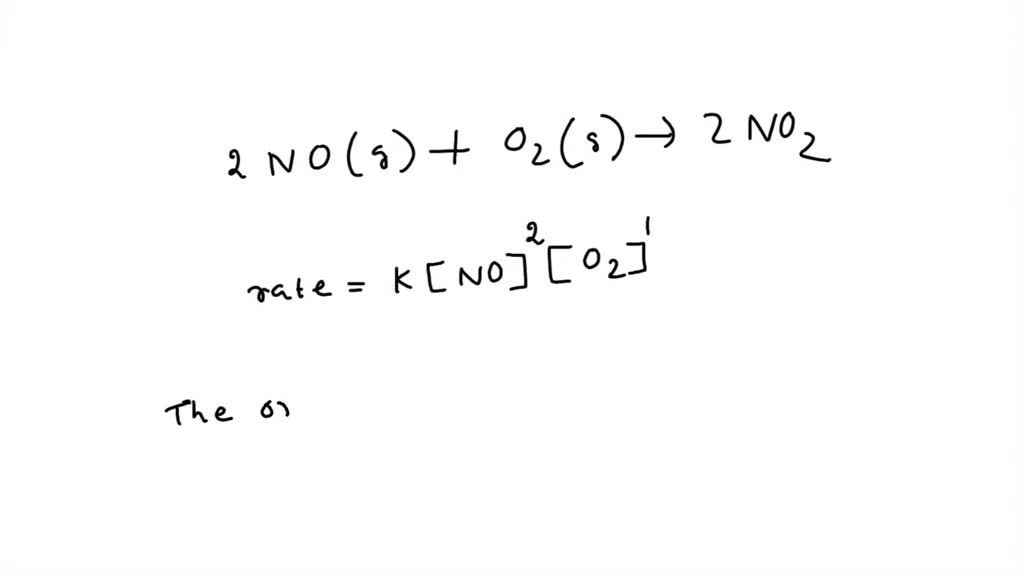

Related Post

<font face="Minecraft Font">UNLOCKING CREATIVITY: THE EVOLUTION OF MINECRAFT FONT GENERATOR</font>

24 Hour Booking Metro Jail Mobile Al: A Comprehensive Guide to Understanding the Process and Facility

The Rise of J Arcive: Revolutionizing Digital Data Storage and Management

The Shocking Truth Behind YNW Melly's Net Worth: From Humble Beginnings to Lavish Lifestyle

