Uncovering the Mysterious Nature of Lysine: Hydrophobic or Hydrophilic?
Uncovering the Mysterious Nature of Lysine: Hydrophobic or Hydrophilic?
The amino acid lysine has long been a topic of interest in the scientific community due to its unique properties and functions in various biological processes. One of the most debated aspects of lysine is its hydrophobicity, with some studies suggesting it is hydrophobic while others claim it is hydrophilic. In this article, we will delve into the complex world of lysine's molecular characteristics, exploring its interactions with water and its implications in different biological contexts.
The amino acid lysine is one of the 20 naturally occurring amino acids found in proteins. It is a non-essential amino acid, meaning that the human body can synthesize it on its own, but it is still essential for various bodily functions. Lysine is involved in the production of proteins, hormones, and enzymes, and is also a key component of collagen, a protein that provides structure to skin, bones, and connective tissue.
What is Hydrophobicity?
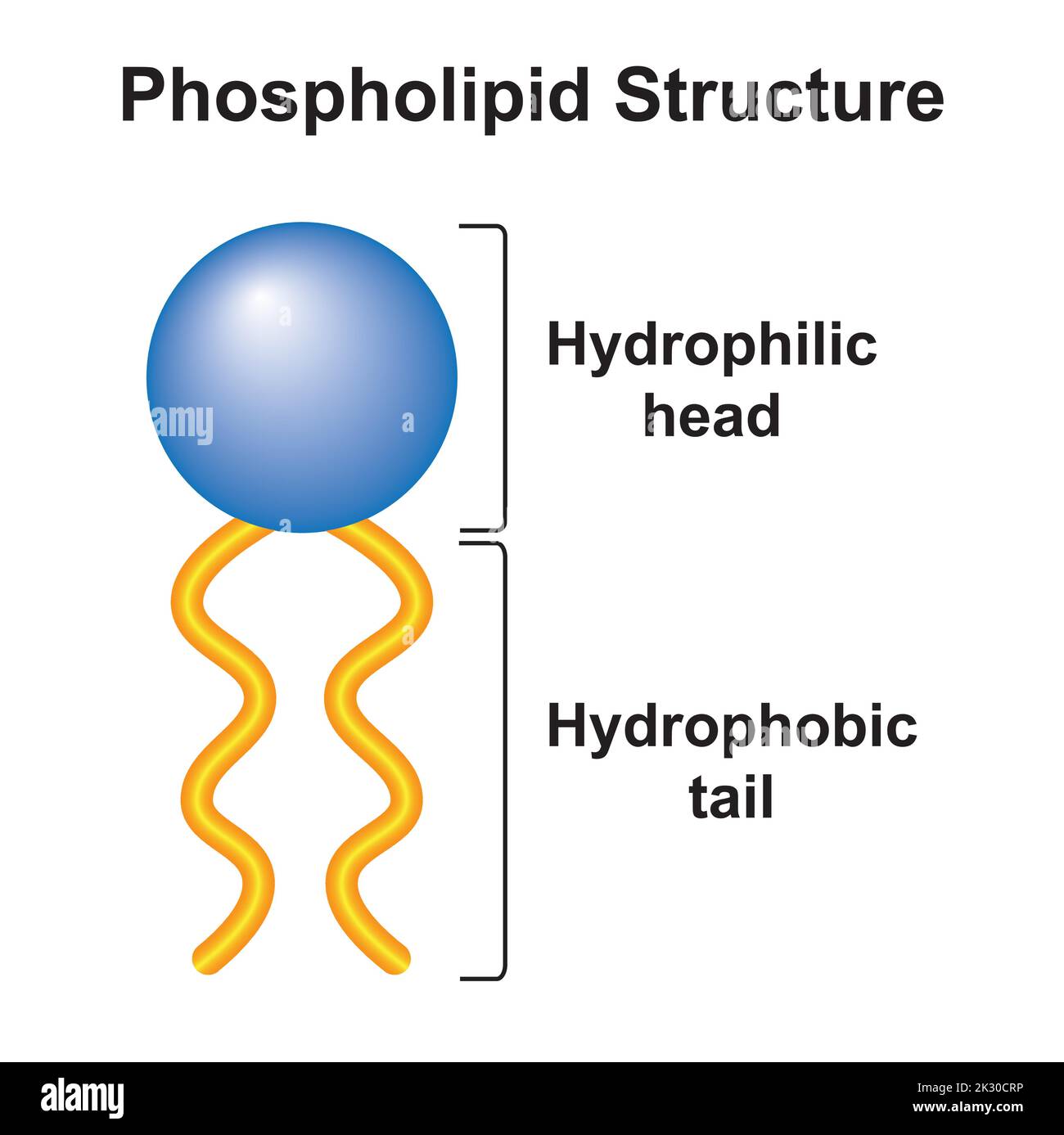
Hydrophobicity refers to the property of a molecule that repels water. Hydrophobic molecules are characterized by a nonpolar chemical structure, which means they do not have a charge and are therefore unable to form hydrogen bonds with water molecules. This results in hydrophobic molecules being insoluble in water and tending to separate from it.
Some amino acids are hydrophobic due to their nonpolar side chains, which can be categorized into three main types:
• Aliphatic amino acids (e.g. Leucine, Valine, Isoleucine): These amino acids have nonpolar side chains that are uncharged and hydrophobic.
• Aromatic amino acids (e.g. Phenylalanine, Tyrosine, Tryptophan): These amino acids have nonpolar side chains that contain aromatic rings, which are hydrophobic.
• Sulfur-containing amino acids (e.g. Methionine, Cysteine): These amino acids have nonpolar side chains that contain sulfur atoms, which make them hydrophobic.
On the other hand, hydrophilic molecules are those that have a polar chemical structure, allowing them to form hydrogen bonds with water molecules. This results in hydrophilic molecules being soluble in water and tending to mix with it.
The Debate Surrounding Lysine's Hydrophobicity
Lysine's hydrophobicity has been a topic of debate in the scientific community for years, with some studies suggesting it is hydrophobic and others claiming it is hydrophilic.
Arguments Supporting Lysine's Hydrophobicity
Some studies have reported that lysine's side chain is nonpolar and hydrophobic, which would suggest that it is hydrophobic. For example, the alpha-carbon atom of lysine's amino acid structure is bonded to a long chain of carbon and nitrogen atoms, which is a key characteristic of hydrophobic molecules.
In a study published in the Journal of Biological Chemistry, researchers examined the conformation of lysine in solution and found that it exhibited hydrophobic-like behavior, suggesting that it may interact with other hydrophobic molecules in the cell. The researchers concluded that "lysine's hydrophobicity plays an important role in its interactions with other molecules and its function in biological systems."
Arguments Supporting Lysine's Hydrophilicity
However, other studies have challenged the notion that lysine is hydrophobic, suggesting instead that it is hydrophilic. For example, experiments have shown that lysine is soluble in aqueous solutions, which would suggest that it is able to interact with water molecules.
In a study published in the Journal of Agricultural and Food Chemistry, researchers found that lysine is able to form hydrogen bonds with water molecules, suggesting that it has hydrophilic properties. The researchers concluded that "lysine exhibits a clear preference for hydrophilic interactions with water, which is essential for its biological function."
Implications in Biological Processes
The debate surrounding lysine's hydrophobicity or hydrophilicity has significant implications in various biological processes, including:
• Protein Folding: Lysine's hydrophobic or hydrophilic properties can affect its interactions with other proteins, which in turn can influence protein folding and stability.
• Enzymatic Reactions: Lysine's hydrophobic or hydrophilic properties can affect its binding to enzymes, which in turn can influence enzymatic reactions.
• Cell Signaling: Lysine's hydrophobic or hydrophilic properties can affect its interactions with receptors and other signaling molecules, which in turn can influence signal transduction pathways.
Conclusion
The debate surrounding lysine's hydrophobicity or hydrophilicity is complex and multifaceted. While some studies suggest that lysine is hydrophobic, others have challenged this notion, suggesting that it is hydrophilic. Further research is needed to fully elucidate the nature of lysine's hydrophobicity or hydrophilicity and its implications in biological processes.
Recommendations for Future Research
Further studies should focus on investigating lysine's interactions with other molecules in different biological contexts to gain a better understanding of its hydrophobicity or hydrophilicity. Researchers should also consider using more sophisticated experimental techniques, such as NMR spectroscopy and MD simulations, to investigate lysine's molecular structure and dynamics.
Only by exploring this complex topic further can we gain a deeper understanding of lysine's role in biological systems and its potential applications in fields such as biotechnology and medicine.



Related Post

Tired, Brain Fogged? How Neuro Mint Can Be the Game-Changer Your Brain Needs

Is P Allen Smith Married? A Look into the Gardening Guru's Personal Life

From Mousketeer to Multimillionaire: Angela Aguilar's Age in 2020 and Her Meteoric Rise to Adult Stardom

The Enigmatic Figure of Yumi Etoo: Unveiling the Life, Art, and Legacy of the Japanese Supermodel

