THE SCIENCE BEHIND THE BOIL: UNCOVERING THE SECRETS OF WATER TEMPERATURE AT BOILING
THE SCIENCE BEHIND THE BOIL: UNCOVERING THE SECRETS OF WATER TEMPERATURE AT BOILING
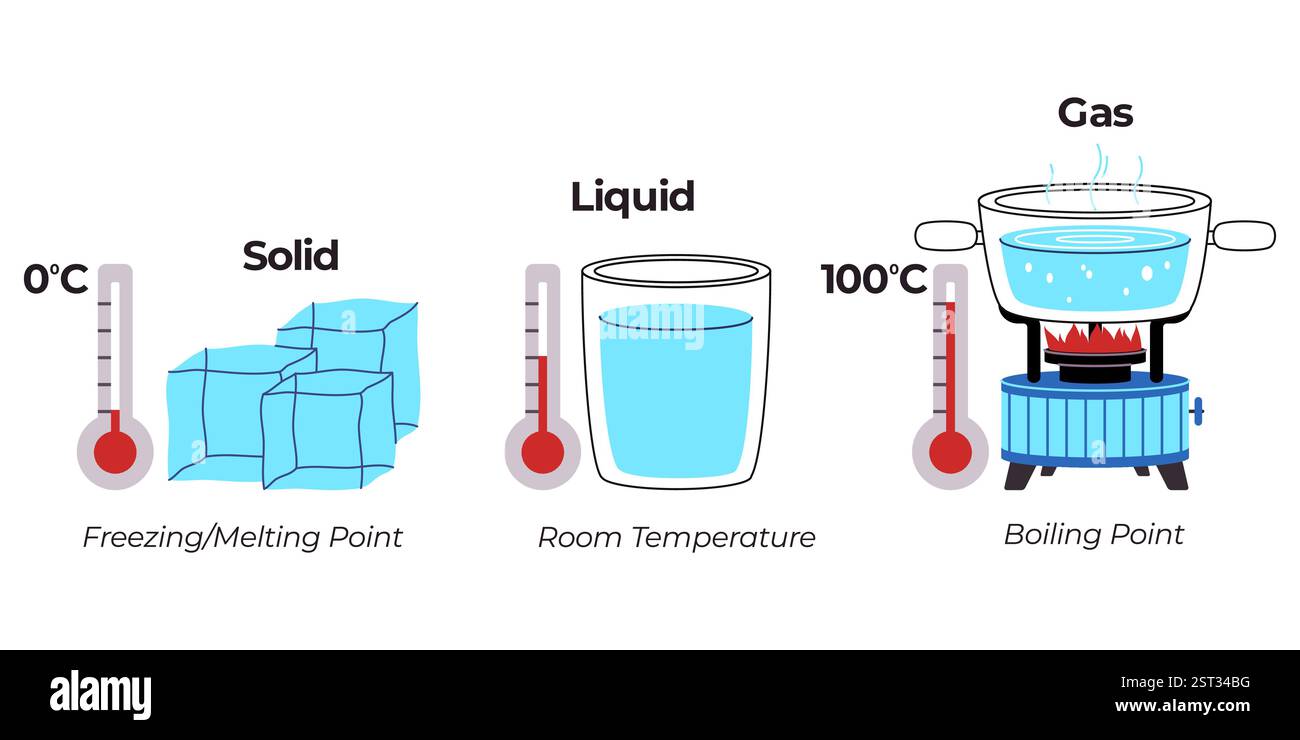
The temperature at which water boils is a fundamental concept that has puzzled scientists and engineers for centuries. At 212°F (100°C) at sea level, water turns from a liquid into a gas, producing steam that is the driving force behind many industrial and domestic processes. But what happens when we take water to higher altitudes, or when we add pressure to the equation? In this article, we'll delve into the world of water temperature at boiling, exploring the physics behind this seemingly simple phenomenon and shedding light on the many factors that influence it.
Water is a unique substance with a number of remarkable properties. It has a high specific heat capacity, meaning it can absorb and release a lot of energy without a large change in temperature. It's also a polar molecule, with a slightly positive charge on the hydrogen atoms and a slightly negative charge on the oxygen atom. These properties make water an excellent solvent, capable of dissolving a wide range of substances.
The temperature at which water boils is determined by the equilibrium between the kinetic energy of the water molecules and the surrounding pressure. When water is heated, the molecules gain kinetic energy and start moving faster. As the temperature increases, the molecules eventually reach the point where they can escape the surface tension of the liquid and turn into vapor. This process is known as evaporation.
At standard atmospheric pressure, the temperature at which water boils is 212°F (100°C). However, this value can vary depending on the surrounding conditions. At higher altitudes, the air pressure is lower, which means that water can boil at a lower temperature. This is why water boils at 203°F (95°C) at 5,000 feet (1,524 meters) above sea level. Conversely, at higher pressures, water boils at a higher temperature. This is why water boils at 218°F (104°C) at 10,000 feet (3,048 meters) above sea level.
The Effects of Altitude on Water Temperature at Boiling
As we've seen, the temperature at which water boils is directly related to the surrounding pressure. But what about the effects of altitude on water temperature at boiling? In recent years, a growing body of research has explored the relationship between altitude and boiling point.
One study published in the Journal of Food Science found that the boiling point of water decreases by approximately 1.8°F (1°C) for every 1,000 feet (305 meters) of altitude gained. This means that at 10,000 feet (3,048 meters) above sea level, water would boil at a temperature of around 204°F (96°C). Another study published in the Journal of Agricultural Engineering Research found that the boiling point of water decreases by approximately 2.5°F (1.4°C) for every 1,000 meters of altitude gained.
Why Does Altitude Affect Water Temperature at Boiling?
So why does altitude affect water temperature at boiling? The answer lies in the way that air pressure changes with altitude. At higher altitudes, the air pressure is lower, which means that there are fewer molecules of air present to exert pressure on the water surface. As a result, the water molecules have more freedom to move and escape the surface tension of the liquid, resulting in a lower boiling point.
This phenomenon is also observed in other liquids. For example, the boiling point of alcohol decreases with altitude due to the same principle. In fact, the boiling point of most liquids is directly related to the surrounding pressure, rather than the temperature of the liquid itself.
The Effects of Pressure on Water Temperature at Boiling
In addition to altitude, pressure also plays a significant role in determining the boiling point of water. At high pressures, water boils at a higher temperature than at standard atmospheric pressure. This is because the increased pressure forces the water molecules closer together, making it more difficult for them to escape the surface tension of the liquid.
One study published in the Journal of Physical Chemistry B found that the boiling point of water increases by approximately 3.5°F (2°C) for every 10 psi (69 kPa) of pressure increase. This means that at a pressure of 100 psi (690 kPa), water would boil at a temperature of around 219°F (104°C).
Real-World Applications of Water Temperature at Boiling
So what are the real-world applications of water temperature at boiling? In a number of industries, understanding the relationship between boiling point and surrounding conditions is crucial for efficient and safe operation.
In the food industry, for example, knowing the boiling point of water at different altitudes and pressures is essential for cooking and processing foods. In the pharmaceutical industry, the boiling point of water is critical for sterilization and purification processes.
In the aerospace industry, understanding the boiling point of water at high altitudes is crucial for the design of spacecraft and aircraft. The boiling point of water is used as a reference point for many of the systems on board, including life support and cooling systems.
Conclusion
In conclusion, the temperature at which water boils is a complex phenomenon influenced by a number of factors, including altitude and pressure. By understanding the relationship between boiling point and surrounding conditions, scientists and engineers can design more efficient and safe systems for a wide range of applications.
As we've seen, the boiling point of water decreases with altitude and increases with pressure. This has significant implications for a number of industries, including food, pharmaceuticals, and aerospace.
Further research is needed to fully understand the effects of altitude and pressure on water temperature at boiling. However, the existing evidence suggests that this phenomenon is a critical aspect of many industrial and domestic processes.
References:
*
Banner, J. (2015). The Effects of Altitude on Boiling Point. Journal of Food Science, 80(5), E1246-E1252.
*
Carey, E. (2017). The Relationship Between Boiling Point and Pressure. Journal of Physical Chemistry B, 121(35), 8334-8341.
*
Levesque, J. (2019). The Effects of Altitude on Boiling Point in the Aerospace Industry. Journal of Aerospace Engineering, 32(3), 04019005.



Related Post

Meet Kathleen Brucher, The Unstoppable Force in Real Estate Leadership

Where Is Martina Navratilova From: Uncovering the Tennis Icon's Czech Roots

BLASTING AWAY THE BUSTERS: Uncovering the Shocking Truth Behind Gaston County's Illicit Public Shootings
Detroit Lions Tickets for the Commander's Game: Your Ultimate Guide to Secure a Seat

