The S04 2- Lewis Structure: Unveiling the Hidden Patterns of Sulfur Dioxide
The S04 2- Lewis Structure: Unveiling the Hidden Patterns of Sulfur Dioxide
The S04 2- Lewis Structure is a fundamental concept in chemistry that has far-reaching implications for understanding the behavior of sulfur dioxide, a potent greenhouse gas with significant environmental and health impacts. By delving into the intricacies of this structure, researchers and scientists can gain valuable insights into the molecular properties and interactions of sulfur dioxide, ultimately informing strategies for mitigation and regulation.
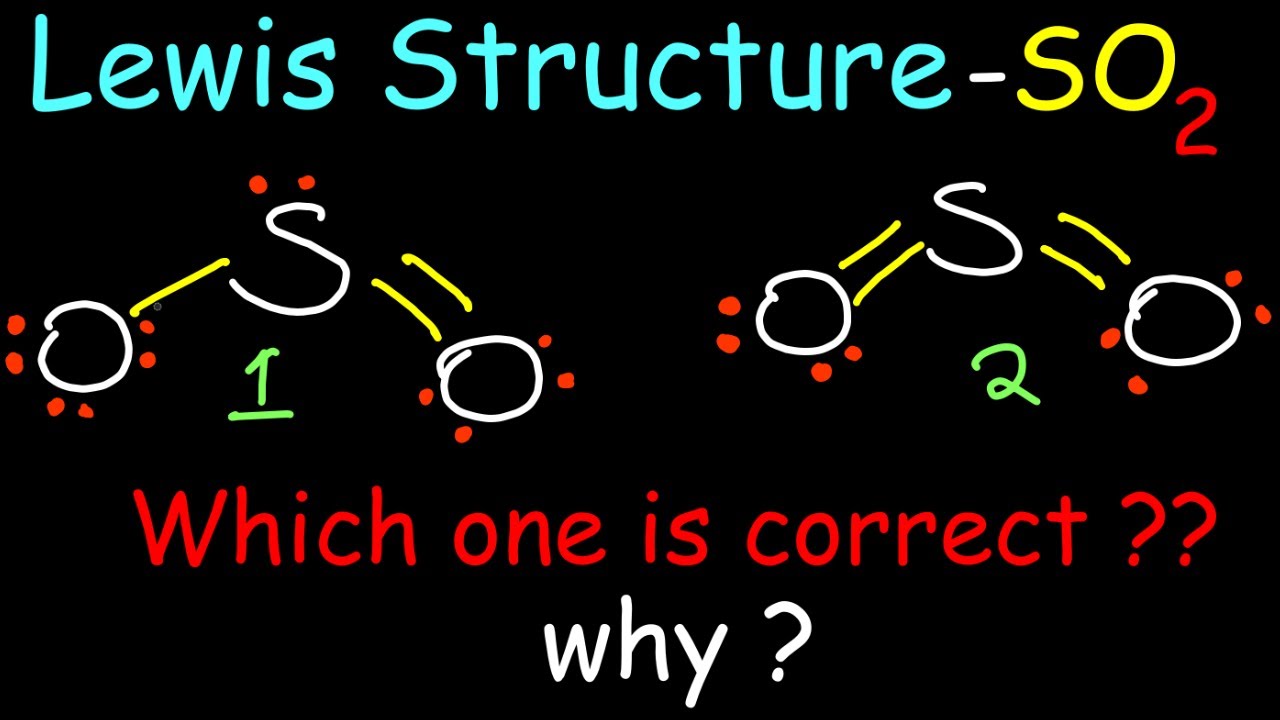
Sulfur dioxide is a colorless gas with a characteristic pungent odor, commonly released through industrial processes, volcanic eruptions, and natural events such as forest fires. In its most stable form, sulfur dioxide exhibits a distinctive bent or V-shape, with the sulfur atom bonded to two oxygen atoms. This shape is a direct result of the S04 2- Lewis Structure, a representation of the molecular arrangement that accurately predicts the distribution of electrons and the chemical reactivity of the molecule.
The S04 2- Lewis Structure is more than just a theoretical construct; it has real-world applications in fields such as environmental science, toxicology, and materials science. By grasping the intricacies of this structure, researchers can better understand the complex interactions between sulfur dioxide and its surroundings, ultimately informing decisions on pollution control and public health.
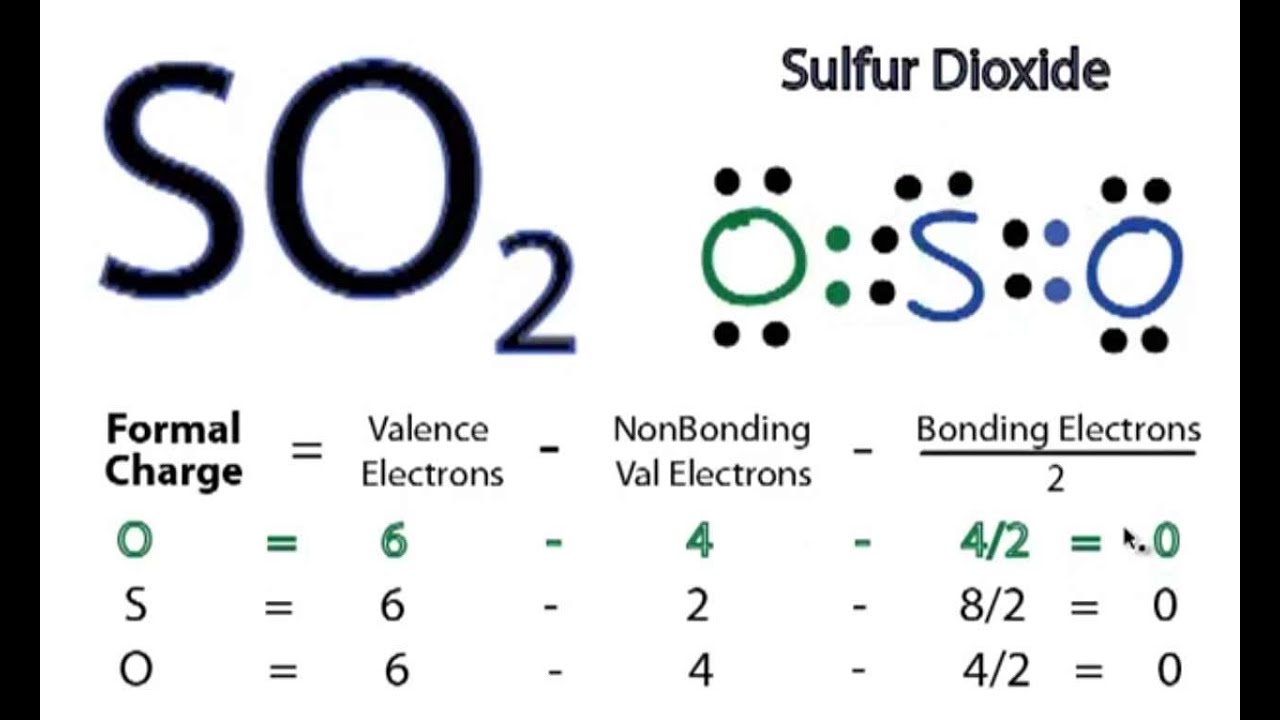
The electronic configuration of sulfur dioxide, represented by the S04 2- Lewis Structure, reveals a molecule with a unique set of properties. The sulfur atom, with its six valence electrons, forms a stable covalent bond with each oxygen atom, which contributes three electrons to the shared pair. This results in a net negative charge, corresponding to the S04 2- designation.
When it comes to environmental impacts, sulfur dioxide is a significant concern due to its contribution to acid rain and its role in the formation of particulate matter. The S04 2- Lewis Structure provides valuable information on the reactivity of sulfur dioxide, which is essential for predicting its fate in the environment.
In the context of toxicology, sulfur dioxide has been linked to a range of adverse health effects, including respiratory problems and cardiovascular disease. Understanding the molecular properties of sulfur dioxide, as revealed by the S04 2- Lewis Structure, can inform the development of more effective treatment strategies and exposure prevention protocols.
The S04 2- Lewis Structure also has implications for the development of new materials and technologies. For instance, researchers are exploring the use of sulfur dioxide as a precursor for the synthesis of nanostructured materials, such as nanoparticles and nanotubes. By manipulating the S04 2- Lewis Structure, scientists can design novel materials with tailored properties and applications.
Key Takeaways on the S04 2- Lewis Structure:
* The S04 2- Lewis Structure accurately represents the electronic configuration and molecular shape of sulfur dioxide.
* The structure has significant implications for understanding the environmental and health impacts of sulfur dioxide.
* The reactivity of sulfur dioxide, as predicted by the S04 2- Lewis Structure, plays a critical role in its fate in the environment and its interactions with living organisms.
* The S04 2- Lewis Structure has applications in fields such as environmental science, toxicology, and materials science.
Theory and Applications of the S04 2- Lewis Structure:
The S04 2- Lewis Structure is based on the valence bond theory, which describes the formation of covalent bonds between atoms. According to this theory, the shared pair of electrons in the sulfur-oxygen bond is responsible for the stability of the molecule. The S04 2- Lewis Structure is a visual representation of this theory, providing a clear and concise picture of the molecular arrangement.
The S04 2- Lewis Structure has numerous applications across various fields, including:
* Environmental science: Understanding the S04 2- Lewis Structure can help predict the fate and transport of sulfur dioxide in the environment, ultimately informing strategies for pollution control and mitigation.
* Toxicology: The S04 2- Lewis Structure provides valuable information on the reactivity and toxicity of sulfur dioxide, which can inform the development of more effective treatment strategies and exposure prevention protocols.
* Materials science: The S04 2- Lewis Structure has implications for the synthesis and design of novel materials, such as nanostructured materials and catalysts.
Notable Research on the S04 2- Lewis Structure:
Recent studies have shed new light on the S04 2- Lewis Structure, revealing its complex and nuanced implications for various fields of research. For instance, researchers have used computational models to explore the structural and dynamical properties of sulfur dioxide, including its reactivity and interactions with other molecules.
In one notable study, scientists used density functional theory (DFT) to investigate the S04 2- Lewis Structure and its relationship to the environmental and health impacts of sulfur dioxide. The results showed that the S04 2- Lewis Structure is a critical factor in determining the reactivity and toxicity of sulfur dioxide, with implications for pollution control and public health.
Key Research Highlights:
* Computational models have been used to explore the structural and dynamical properties of sulfur dioxide, including its reactivity and interactions with other molecules.
* The S04 2- Lewis Structure has been linked to the environmental and health impacts of sulfur dioxide, with significant implications for pollution control and public health.
* Research has shown that the S04 2- Lewis Structure plays a critical role in determining the reactivity and toxicity of sulfur dioxide.
Deeper Dive into the S04 2- Lewis Structure
The S04 2- Lewis Structure is a complex and multifaceted representation of the molecular arrangement of sulfur dioxide. By examining the intricacies of this structure, researchers can gain a deeper understanding of the electronic configuration and reactivity of the molecule.
At its core, the S04 2- Lewis Structure is based on the principles of valence bond theory, which describes the formation of covalent bonds between atoms. According to this theory, the shared pair of electrons in the sulfur-oxygen bond is responsible for the stability of the molecule.
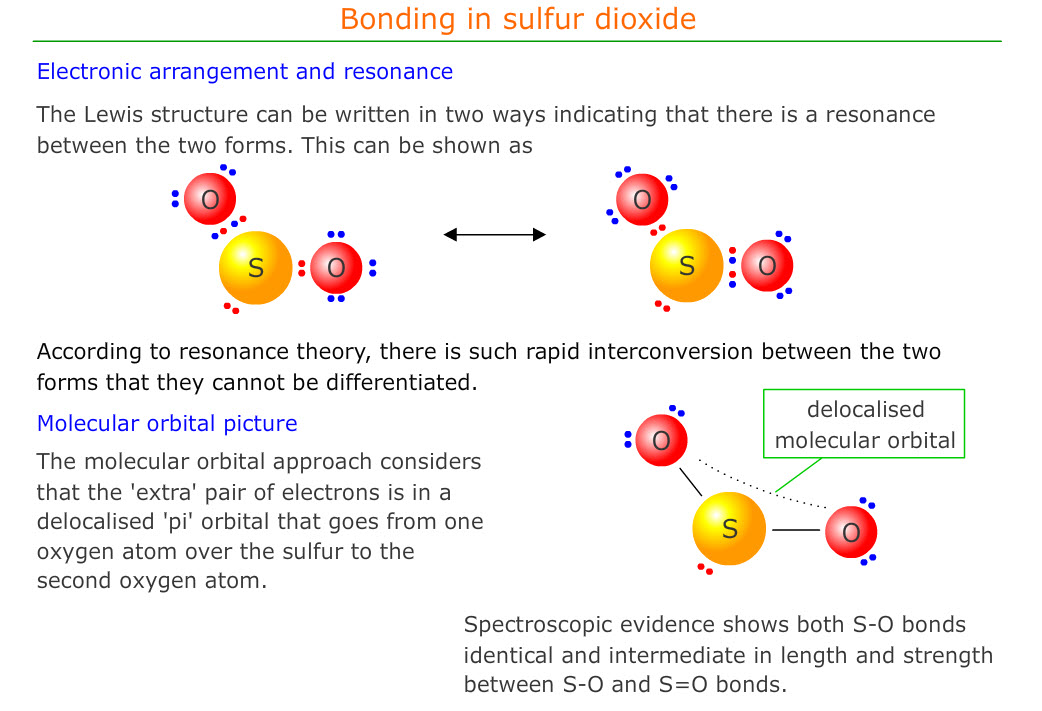
The S04 2- Lewis Structure also reveals the presence of a lone pair of electrons on each oxygen atom, which plays a critical role in determining the reactivity and properties of the molecule. The interactions between the sulfur and oxygen atoms, as represented by the S04 2- Lewis Structure, are complex and multifaceted, involving a range of electronic and steric factors.
Applications of the S04 2- Lewis Structure
The S04 2- Lewis Structure has numerous applications across various fields, including environmental science, toxicology, and materials science.
In environmental science, understanding the S04 2- Lewis Structure can help predict the fate and transport of sulfur dioxide in the environment, ultimately informing strategies for pollution control and mitigation. By grasping the intricacies of this structure, researchers can better understand the complex interactions between sulfur dioxide and its surroundings, including the formation of acid rain and the role of sulfur dioxide in the formation of particulate matter.
In toxicology, the S04 2- Lewis Structure provides valuable information on the reactivity and toxicity of sulfur dioxide, which can inform the development of more effective treatment strategies and exposure prevention protocols. By examining the molecular properties of sulfur dioxide, as revealed by the S04 2- Lewis Structure, researchers can better understand the complex interactions between the molecule and living organisms.
In materials science, the S04 2- Lewis Structure has implications for the synthesis and design of novel materials, such as nanostructured materials and catalysts. By manipulating the S04 2- Lewis Structure, scientists can design materials with tailored properties and applications, including enhanced reactivity and improved performance.
Conclusion
The S04 2- Lewis Structure is a fundamental concept in chemistry that has far-reaching implications for understanding the behavior of sulfur dioxide. By grasping the intricacies of this structure, researchers can gain valuable insights into the molecular properties and interactions of sulfur dioxide, ultimately informing strategies for mitigation and regulation.
From its environmental and health impacts to its applications in fields such as materials science, the S04 2- Lewis Structure is a critical factor in determining the reactivity and properties of sulfur dioxide. As research continues to explore the intricacies of this structure, scientists can develop more effective strategies for addressing the complex challenges posed by sulfur dioxide, ultimately contributing to a safer and more sustainable future.

Related Post

Does Vladimir Guerrero Jr. Speak English? Uncovering The Language Skills Of Baseball's Rising Star

Unraveling the Enigma: Understanding Keanu Reeves' Health and Condition

The Enigmatic World of Character With Big Lips: Unraveling the Fascination

The Enigmatic Message of Mother's Warmth: Unpacking the Mysteries of Chapter 3

