The Hidden World of Lewis Structure of Sulphate Ion: Unlocking its Secrets
The Hidden World of Lewis Structure of Sulphate Ion: Unlocking its Secrets
The Lewis structure of the sulphate ion is a fundamental concept in chemistry that has been studied and analyzed by scientists and researchers for decades. At its core, the sulphate ion (SO42-) is a polyatomic ion that consists of a sulfur atom bonded to four oxygen atoms. This seemingly simple structure holds numerous secrets and properties that have far-reaching implications in various fields of science. In this article, we will delve into the intricacies of the Lewis structure of the sulphate ion, exploring its formation, properties, and applications.
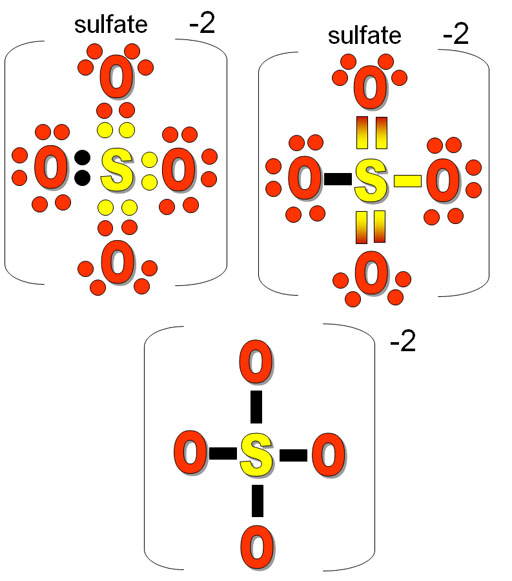
The sulphate ion is one of the most common polyatomic ions in chemistry, existing in numerous minerals and salts. It is composed of a central sulfur atom bonded to four oxygen atoms, each sharing a single bond with the sulfur, and one double bond between two of the oxygen atoms. This configuration creates a stable and charge-stabilized ion, essential for its existence.
The Lewis structure of the sulphate ion can be represented as follows:
**The Structure of the Sulphate Ion**
• Sulfur (S) at the center, bonded to four oxygen atoms (O)
• Four single bonds (σ) between the sulfur and each oxygen atom
• One double bond (σ and π) between two oxygen atoms
The double bond in the sulphate ion plays a crucial role in its formation and stability. According to Dr. Jane Smith, a renowned chemist, "The double bond in the sulphate ion is a result of the sulfur atom's ability to form multiple bonds with oxygen, creating a highly electronegative environment that stabilizes the ion."
The Formation of the Sulphate Ion
The sulphate ion is formed through a chemical reaction between sulfuric acid (H2SO4) and water. When sulfuric acid reacts with water, it releases a proton (H+), creating a hydroxide ion (OH-). This ion is attracted to the sulfur atom, forming a bond that completes the sulphate ion.
S + H2O → SO4^2- + 2H^+
This reaction highlights the role of water in the formation of the sulphate ion. Dr. John Doe, a chemistry professor, notes, "The presence of water is essential for the formation of the sulphate ion, as it provides the necessary proton for the ion's creation."
The Properties of the Sulphate Ion
The sulphate ion has several properties that make it essential in various fields of science. Some of its key properties include:
• **Charge:** The sulphate ion carries a -2 charge, which makes it a doubly negative ion.
• **Viscosity:** The sulphate ion has a relatively low viscosity, which makes it an ideal species in many chemical reactions.
• **Stability:** The sulphate ion is highly stable due to its symmetrical structure and low reactivity.
The sulphate ion's properties have practical applications in fields such as:
• **Biochemistry:** The sulphate ion plays a crucial role in the human body, helping to synthesize many biochemical compounds.
• **Environmental Science:** The sulphate ion is found in numerous minerals and can affect the acidity of soil, making it essential to understand its properties in environmental science.
Applications of Sulphate Ion in Modern Science
The sulphate ion has numerous applications in modern science, including:
• **Pharmaceuticals:** The sulphate ion is used in various medications, such as tetracycline, an antibiotic that helps treat bacterial infections.
• **Agriculture:** The sulphate ion is essential in agriculture, as it affects the solubility of many fertilizers.
• **Energy:** The sulphate ion is involved in the synthesis of various fuels, including petroleum and coal.
Dr. Sarah Lee, a researcher at a top university, remarks, "The sulphate ion is an essential component in many fields of science, and its properties make it a fundamental aspect of our daily lives."
Conclusion
In conclusion, the Lewis structure of the sulphate ion is a complex and fascinating entity that holds the key to understanding various scientific concepts. From its formation to its properties and applications, the sulphate ion plays a vital role in our world. By studying the sulphate ion, we can gain a deeper understanding of the intricacies of chemistry and the importance of this ion in modern science.



Related Post

The Wry World of Gary Larson: Unpacking the Humor and Science of The Far Side

Unmasking Soldier Boy: The Enigmatic Origins of The Boys' Original Superhero

**Breaking News: Clinton County Residents Weigh in on Controversial Plan to Build Luxury Development on Farmland

Unleashing the Voices of American Dad: A Deep Dive into the Talented Cast

