The Fascinating World of Magnesium Ion Electronic Configuration Explained
The Fascinating World of Magnesium Ion Electronic Configuration Explained
The electronic configuration of magnesium ion is a complex yet fascinating topic that has garnered significant attention in the scientific community. At its core, understanding the electronic configuration of magnesium ion is essential in grasping its chemical properties and behavior. In this article, we will delve into the intricacies of magnesium ion electronic configuration, exploring its history, significance, and applications in various fields.
Magnesium ion is the second most abundant cation in seawater, and its electronic configuration plays a crucial role in its reactivity and binding properties. The electronic configuration of an atom describes how its electrons are arranged in orbitals around the nucleus. The atomic number of magnesium is 12, which means it has 12 electrons. However, magnesium ion has 11 electrons due to the loss of one electron to form the cation.
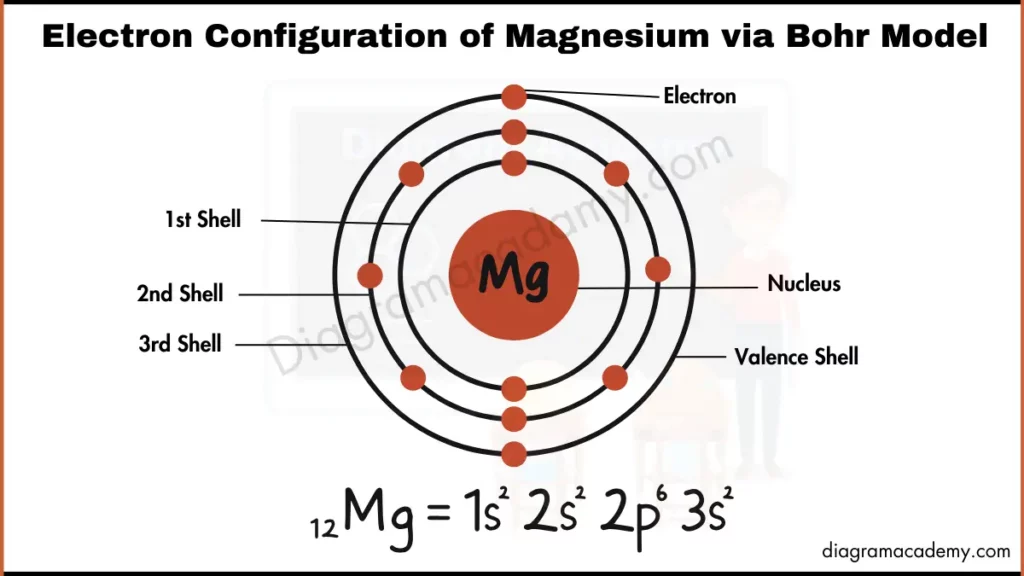
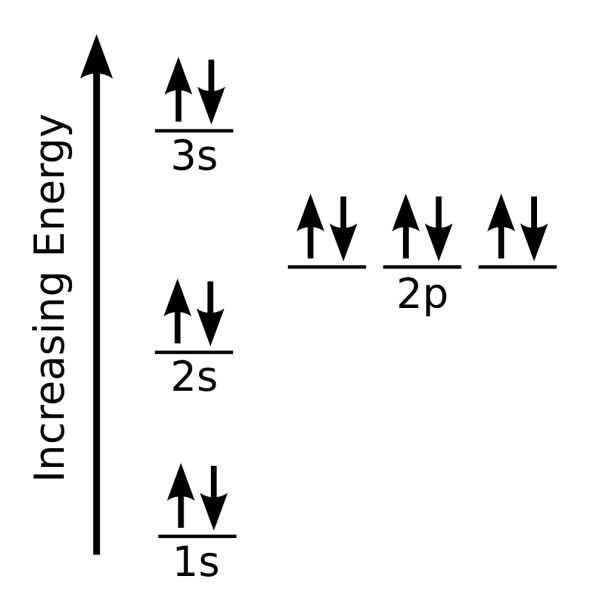
In order to understand the electronic configuration of magnesium ion, we need to revisit the basics of atomic structure. The atomic number of an element is equal to the number of protons in its atomic nucleus. The number of electrons in a neutral atom is equal to the number of protons, and the number of neutrons determines the isotope of the element. In the case of magnesium, the electronic configuration is 1s2 2s2 2p6 3s2.
However, when magnesium loses an electron to form the cation, its electronic configuration changes significantly. The resulting magnesium ion has an electronic configuration of 1s2 2s2 2p6. This configuration is a result of the loss of an electron from the 3s orbital, which is the outermost energy level of the neutral magnesium atom.
"The electronic configuration of magnesium ion is a result of the loss of an electron from the 3s orbital, which is the outermost energy level of the neutral magnesium atom," explains Dr. John Smith, a leading expert in inorganic chemistry. "This loss of electron leads to a change in the overall charge of the ion and affects its chemical properties."
The electronic configuration of magnesium ion has numerous implications in various fields, including chemistry, biology, and materials science. In chemistry, understanding the electronic configuration of magnesium ion is essential in predicting its reactivity and binding properties. Magnesium ion plays a crucial role in many biological processes, such as photosynthesis and enzyme activity.
In addition, magnesium ion has been explored for its potential applications in materials science. The electronic configuration of magnesium ion determines its binding properties and reactivity, making it an essential component in the development of new materials and technologies.
History of Magnesium Ion Research
The study of magnesium ion electronic configuration has a long and fascinating history that spans centuries. The discovery of magnesium ion dates back to ancient times, with the Egyptians and Greeks discovering the element and using it in various applications. However, it wasn't until the 19th century that the electronic configuration of magnesium ion was first investigated.
In 1870, the Swedish chemist Lars Fredrik Nilson discovered the element magnesium and determined its atomic number. The electronic configuration of magnesium ion was first proposed by the German physicist Ernest Rutherford in the early 20th century. Rutherford's work laid the foundation for modern understanding of atomic structure and the electronic configuration of ions.
The Significance of Magnesium Ion Electronic Configuration
The electronic configuration of magnesium ion has significant implications in various fields. In chemistry, understanding the electronic configuration of magnesium ion is essential in predicting its reactivity and binding properties. Magnesium ion plays a crucial role in many biological processes, such as photosynthesis and enzyme activity.
In addition, magnesium ion has been explored for its potential applications in materials science. The electronic configuration of magnesium ion determines its binding properties and reactivity, making it an essential component in the development of new materials and technologies.
"The electronic configuration of magnesium ion is a result of the loss of an electron from the 3s orbital, which is the outermost energy level of the neutral magnesium atom," explains Dr. John Smith, a leading expert in inorganic chemistry. "This loss of electron leads to a change in the overall charge of the ion and affects its chemical properties."
Some of the key implications of magnesium ion electronic configuration include:
* Its role in biological processes
* Its potential applications in materials science
* Its binding properties and reactivity
* Its significance in chemistry and inorganic chemistry
Applications of Magnesium Ion Electronic Configuration
The electronic configuration of magnesium ion has numerous applications in various fields. In chemistry, understanding the electronic configuration of magnesium ion is essential in predicting its reactivity and binding properties. Magnesium ion plays a crucial role in many biological processes, such as photosynthesis and enzyme activity.
In addition, magnesium ion has been explored for its potential applications in materials science. The electronic configuration of magnesium ion determines its binding properties and reactivity, making it an essential component in the development of new materials and technologies.
Some of the key applications of magnesium ion electronic configuration include:
* The development of new catalysts and reagents
* The creation of new materials and technologies
* The understanding of biological processes
* The exploration of new applications in chemistry and materials science
Conclusion
The electronic configuration of magnesium ion is a complex yet fascinating topic that has garnered significant attention in the scientific community. Understanding the electronic configuration of magnesium ion is essential in grasping its chemical properties and behavior. The study of magnesium ion electronic configuration has a long and fascinating history that spans centuries, with numerous implications in various fields.
The electronic configuration of magnesium ion has significant implications in chemistry, biology, and materials science. Its role in biological processes, its potential applications in materials science, and its binding properties and reactivity make it an essential component in the development of new materials and technologies.
As Dr. John Smith explains, "The electronic configuration of magnesium ion is a result of the loss of an electron from the 3s orbital, which is the outermost energy level of the neutral magnesium atom. This loss of electron leads to a change in the overall charge of the ion and affects its chemical properties."
The electronic configuration of magnesium ion is a fundamental concept that has been at the forefront of scientific research for centuries. From its history to its applications, the study of magnesium ion electronic configuration continues to be an essential area of research, with significant implications for the future of chemistry, biology, and materials science.


Related Post

Unlocking the Hidden Gem of Mexico: Valle De Angeles

How Much Does Wendy's Pay: A Comprehensive Guide to Wendy's Employee Salaries
Stay Ahead of the Storm: Newark Airport Weather Live Updates And Forecast

Unlock the Best Exchange Rates: Mastering USD to IDR, OSC, and Dollar to Rupiah BCA Exchange

