Sn1 Reaction Examples
The world of organic chemistry is a vast and complex landscape, with countless reactions and mechanisms waiting to be unraveled. Among these, the Sn1 reaction stands out as a crucial example of a stoichiometric nucleophilic substitution reaction, where one type of molecule is substituted for another under mild conditions. In this article, we'll delve into the world of Sn1 reactions, exploring what they are, how they work, and providing numerous examples to illustrate the concept.
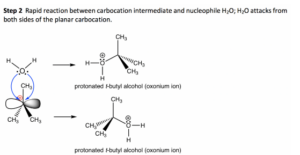
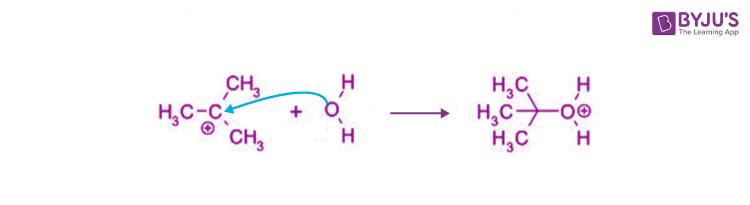
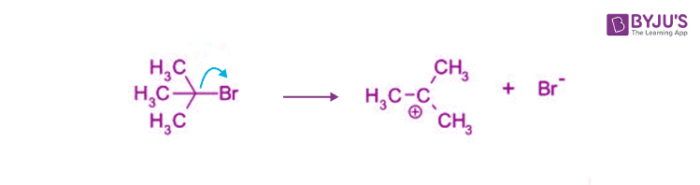
The Sn1 reaction is characterized by a rate-determining step, where the leaving group departs from the substrate, forming a carbocation intermediate. This highly reactive species is then attacked by a nucleophile, resulting in the formation of a new bond. It's a reaction that requires patience and understanding, but yields profound results.
Synthetic organic chemists rely heavily on Sn1 reactions, which are not only a critical building block in the synthesis of complex molecules but also a key to understanding the fundamental principles of organic chemistry. By examining Sn1 reactions, we can gain insight into the intricacies of molecular interactions and design efficient synthesis strategies.
Understanding the Sn1 Reaction Mechanism
The Sn1 reaction involves a single-step process where the leaving group departs from the substrate, forming a carbocation intermediate. This carbocation can then be attacked by a nucleophile, resulting in the formation of a new bond. This process can be represented by a few key steps:
Factors Affecting the Sn1 Reaction Rate
The rate of the Sn1 reaction can be influenced by several key factors. These include:
Sn1 Reaction Examples
Here are some examples of Sn1 reactions:
Example 1: Propan-2-yl bromide Sn1 reaction
In this example, the propan-2-yl bromide undergoes an Sn1 reaction with hydroxide as the nucleophile. The hydroxide ion attacks the carbocation intermediate, resulting in the formation of methanol and acetone.

Example 2: Toluene-4-sulfonic acid Sn1 reaction
In this example, the toluene-4-sulfonic acid undergoes an Sn1 reaction in the presence of a strong base. The resulting product is the toluene-4-sulfinate anion, which can be converted into other useful compounds.

From these examples, it is clear that the Sn1 reaction plays a crucial role in the synthesis of complex molecules. As we delve deeper into the fundamental principles of organic chemistry, the importance of Sn1 reactions becomes increasingly apparent.
Conclusion
The Sn1 reaction is a powerful tool in the world of organic chemistry. Its impact extends far beyond the realm of synthetic chemistry, providing a glimpse into the intricate dance of molecular interactions. By mastering Sn1 reactions, chemists can unlock new avenues for the synthesis of complex molecules, opening the door to a world of innovative discoveries and applications. Whether it's the synthesis of therapeutics, the development of new materials, or the exploration of fundamental principles, the Sn1 reaction stands as a cornerstone in the world of organic chemistry.

Related Post

The Next Generation of Business Leaders: David Rubenstein's Children Meet Gabrielle Rubenstein And Alexa Rachlin

Meet Paul Thomas: The Husband Behind Clevelands Popular Weather Personality Betsy Kling

The Unseen Benefits of Amy Lynn's Devotion to Data Science: Unlocking the Power of Insight

**"Hoops in the Bay: Tampa's Thriving Basketball Scene and Pro Options"**

